science chemistry experiment states of matter Fundamental Photographs The Art of Science

Phase diagram with a triple point O of water analogy. Download Scientific Diagram
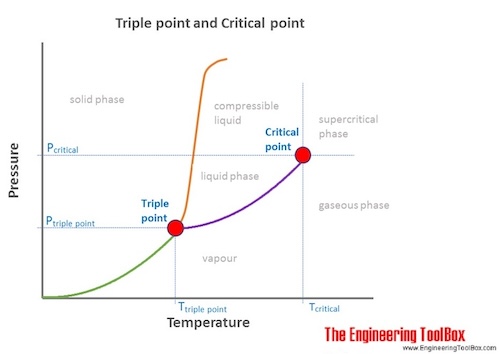
Triple point of water. In chemistry and physics, the triple point is the temperature and pressure at which solid, liquid, and vapor phases of a particular substance coexist in equilibrium. It is a specific case of thermodynamic phase equilibrium. The term "triple point" was coined by James Thomson in 1873.

Why use a triple point of water? Namibian Mining News
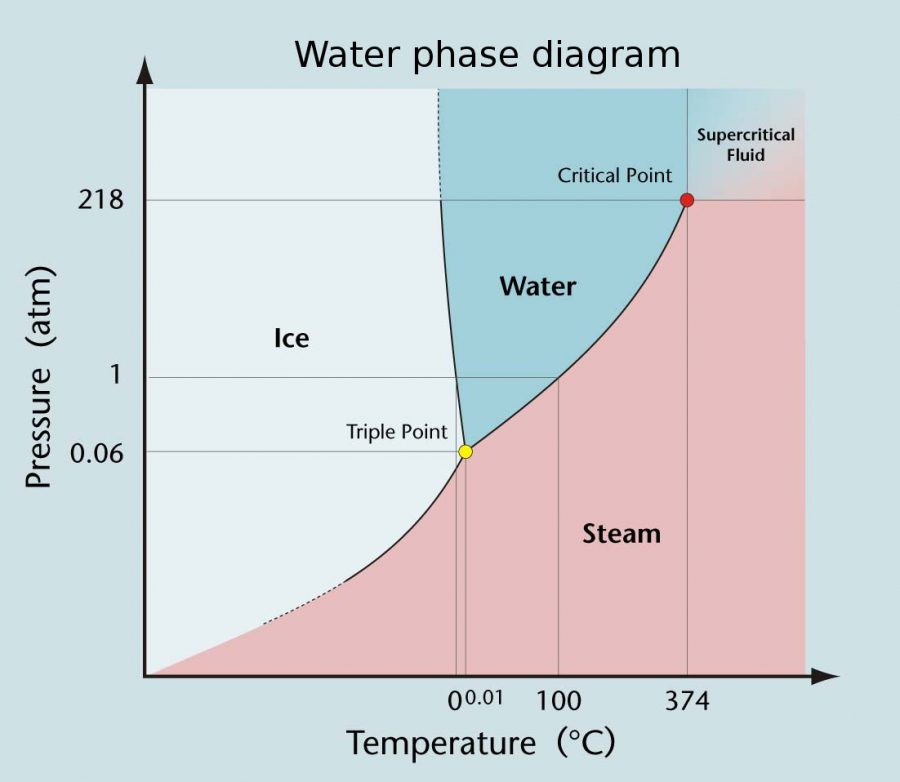
This "triple point" is different for each substance. For example, the triple point of water is reached when the temperature is 273.16 kelvins (or 0.01 °C) and the pressure is at 0.006 atmospheres, while the triple point of carbon dioxide requires the temperature of −56.6 °C and pressure of 5.11 atm. Because of the limited conditions in.

Triple point of water YouTube
The triple point occurs where the solid, liquid, and gas transition curves meet. The triple point is the only condition in which all three phases can coexist, and is unique for every material. Water reaches its triple point at just above freezing (0.01° C) and at a pressure of 0.006 atm. Related reading: Thermodynamic equilibrium.
[DIAGRAM] Test Point Diagram 161.35.132.113
The triple point of water is at 273.16 kelvin (0.01 °C or 32.02 °F) and a pressure of 611.7 pascals (6.117 millibars, 0.0060373057 atm). (Some sources list a pressure of 611.73 Pa, while others cite a pressure of 611.657 Pa.) At this point, ice, water, and water vapor coexist in stable form. The tiniest change in temperature or pressure.

Thermodynamics Explaining the Triple Point YouTube
The triple point occurs where the solid, liquid, and gas transition curves meet. The triple point is the only condition in which all three phases can coexist.

What is Tripple point? PhysicStuff
The triple point of water is important enough to merit its own line: T3 = 273.1K. This is equivalent to 0.01oC and 32.02oF, and thus we use T3 to help set the freezing point of water. Absolute zero is defined as being equivalent to that of 0 Kelvin, which is -273.15oC and and -459.67oF, and this is a standard that is accepted internationally.

Preparation of Equipment for the Realisation of Water Triple Point Cells YouTube
The triple point occurs where the solid, liquid, and gas transition curves meet. The triple point is the only condition in which all three phases can coexist, and is unique for every material. Water reaches its triple point at just above freezing (0.1° C) and at a pressure of 0.006 atm.

Triple Point of Water YouTube
The triple point of water corresponds to the minimum pressure at which water in the liquid state can exist. At pressures below the triple point (as in outer space), solid ice when heated at constant pressure is converted directly into water vapor in a process known as sublimation. In general, sublimation is a phase change of a substance.
The Triple Point of Water Coexistence of Solid Liquid and Gas
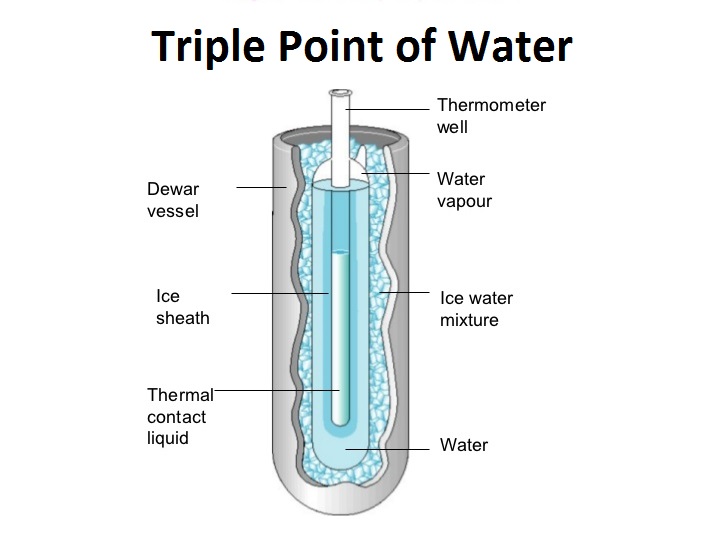
This experiment demonstrates the triple point of a substance. Watch how water behaves at the triple point where it co-exists in solid, liquid and vapour form.
Triple Point of Water The Temperature Where All Three Phases Coexist NA Eye
The triple point of a substance is the temperature and pressure at which the three phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium. The triple point of water is used to define the Kelvin (K), the base unit of thermodynamic temperature in the International System of Units (SI).

Triple point of water analogy. intensive variables critical... Download Scientific
eCHEM 1A: Online General ChemistryCollege of Chemistry, University of California, Berkeleyhttp://chemistry.berkeley.edu/echem1aCurriculum and ChemQuizzes dev.

science chemistry experiment states of matter Fundamental Photographs The Art of Science
Difference between Boiling point and Triple point. In the case of a triple point, all the states of matter exist while in the case of a boiling point only two states of matter exist. It is impossible to drop the boiling point below the triple point. Suggest Corrections. 6.

Triple Point Definition Triple Point of Water
Examples of Triple Point in Nature. One example of a triple point in nature is the triple point of water. At this point, water can exist in all three states: ice, liquid, and vapor. The triple point of water occurs at a temperature of 0.01°C and a pressure of 611.73 pascals. This is why, at high altitudes, water can boil at a lower temperature.

Triple Point Phase Diagram of water YouTube
A typical phase diagram.The solid green line applies to most substances; the dashed green line gives the anomalous behavior of water. In thermodynamics, the triple point of a substance is the temperature and pressure at which the three phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium. It is that temperature and pressure at which the sublimation, fusion.

Triple Point of Water YouTube
This paper is a part of guidelines, prepared on behalf of the Consultative Committee for Thermometry, on the methods how to realize the International Temperature Scale of 1990. It discusses all major issues linked to the triple point of water when used as a fixed point for the realization of the kelvin. 1. Introduction.

Triple point of water analogy. intensive variables critical... Download Scientific
Triple point of water is 273 K temperature and 0.46 cm of mercury pressure. The triple point of water, T 3 = 273.16 K, is the standard fixed-point temperature for the calibration of thermometers. This agreement also sets the size of the kelvin as 1/273.16 of the difference between the triple-point temperature of water and absolute zero.