Bromine (Br) AMERICAN ELEMENTS

How Can We Find A Electron Configuration For Bromine (Br)
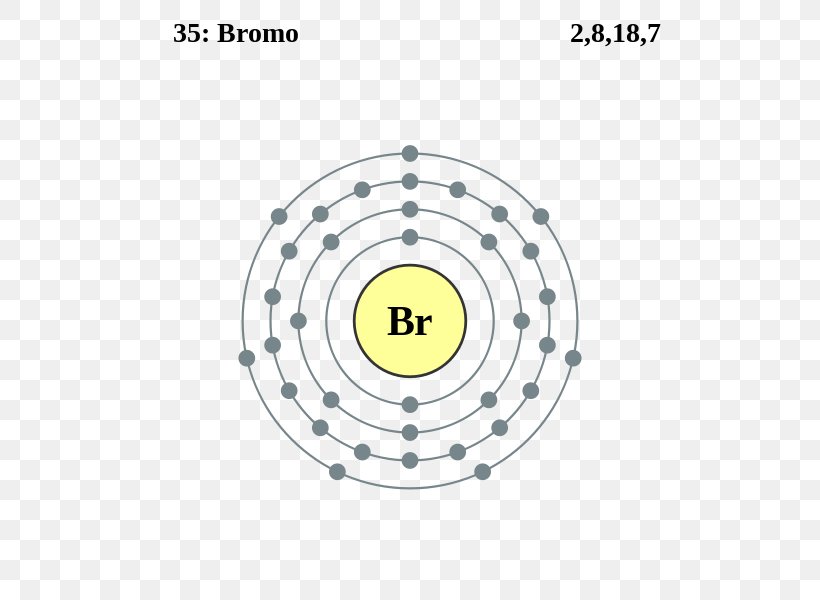
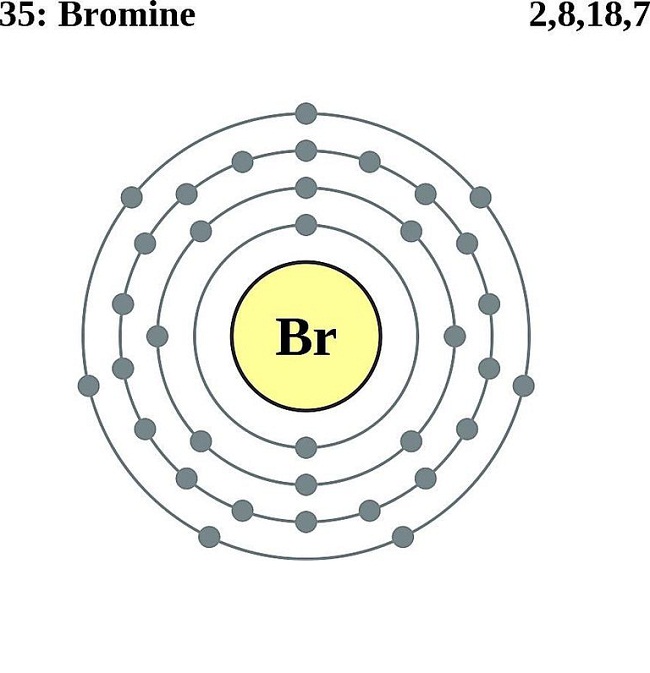
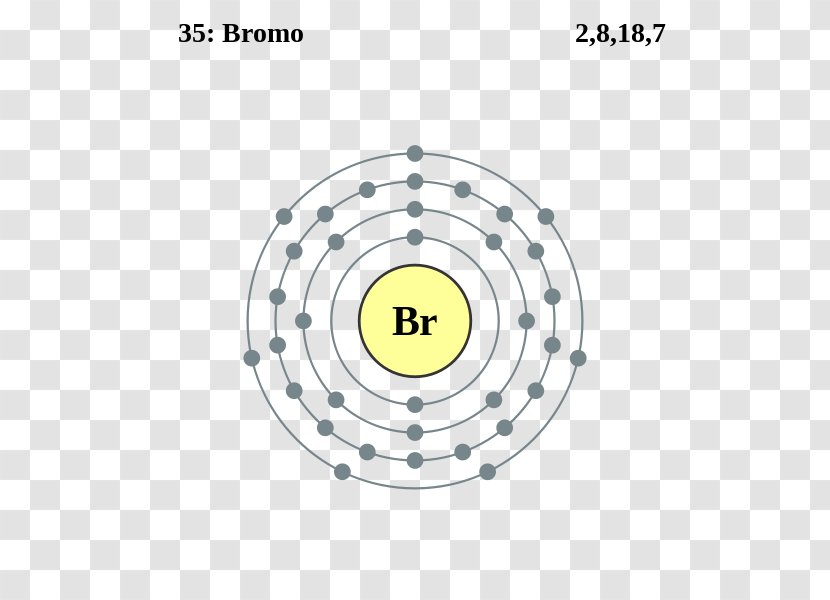
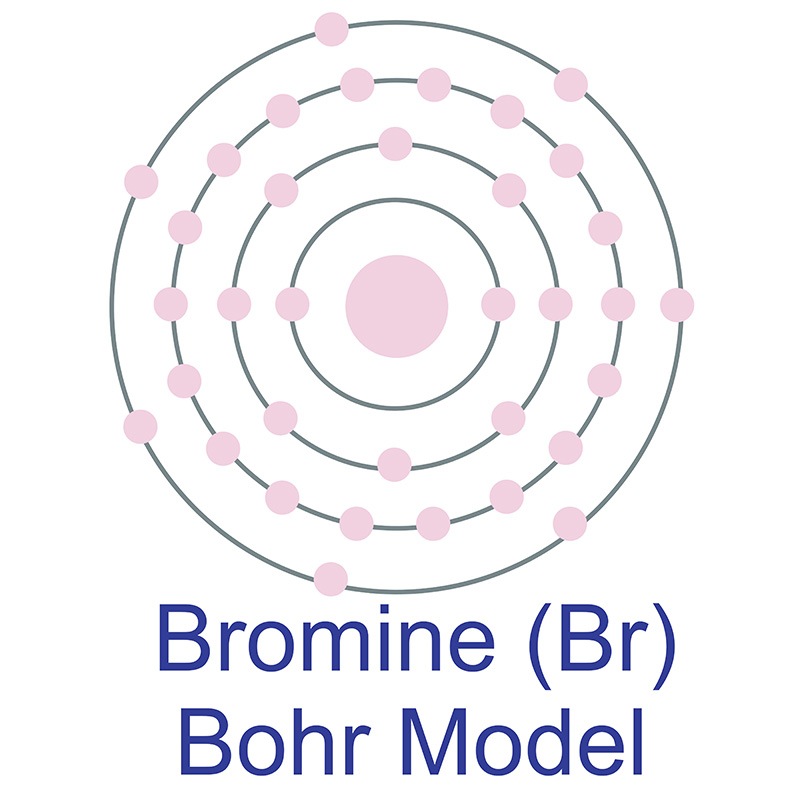
The Bohr Model of Bromine (Br) has a nucleus with 45 neutrons and 35 protons. This nucleus is surrounded by four electron shells. The first shell of the Bohr diagram of Bromine has 2 electrons, the 2nd shell has 8, the 3rd shell has 18, and the 4th shell has 7 electrons. Also check - How to draw Bohr model diagram for an atom?

Bromine (Br) Properties & Uses StudiousGuy
An electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. Electrons are represented by dots or crosses and are positioned in energy levels, or 'shells', around the central nucleus. This is sometimes called the Bohr, or the 'solar system', model.
Electron Configuration For Bromine
Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. Figure 2 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. The shell closest to the nucleus is.

Bromine Formula \({\rm{B}}{{\rm{r}}_2}\) Structure, Molar Mass, IUPAC
Bohr model is a structural model in which the negatively charged electrons revolve around the positively charged nucleus. This is similar to the planets revolving around the sun, except that the orbits are non-planar. The electrons move in a fixed orbits (shells) and each orbit has a fixed energy. Each orbit (or shell) can hold a certain number.
Bromine Facts, Symbol, Discovery, Properties, Uses
The Bohr Model of Bromine (Br) has a nucleus that contains 45 neutrons and 35 protons. This nucleus is surrounded by four-electron shells named K-shell, L-shell, M-shell, and N-shell. The outermost shell in the Bohr diagram of Bromine contains 7 electrons that also called valence electrons. Page Contents show How to draw Bohr Model of Bromine (Br)?

Bromine Bohr Diagram
Bromine (Br) electron configuration (Bohr model) Electron configuration through orbitals follows different principles. For example Aufbau principle, Hund's principle, and Pauli's exclusion principle. Bromine atom electron configuration through orbit Scientist Niels Bohr was the first to give an idea of the atom's orbit.

Bohr Model Bromine Atom Electron Structure Stock Vector (Royalty Free
0:00 / 3:51 Bohr-Rutherford Diagram for Bromine (Br) chemistNATE 251K subscribers Subscribe 230 17K views 3 years ago Bromine has 18 electrons in its third shell because it is past Zinc on the.
Electron Configuration Bromine Chemical Element Shell Bohr Model
In 1913, a Danish physicist, Niels Bohr (1885-1962; Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. Bohr's model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii.

7+ bromine bohr diagram RajibRajisha
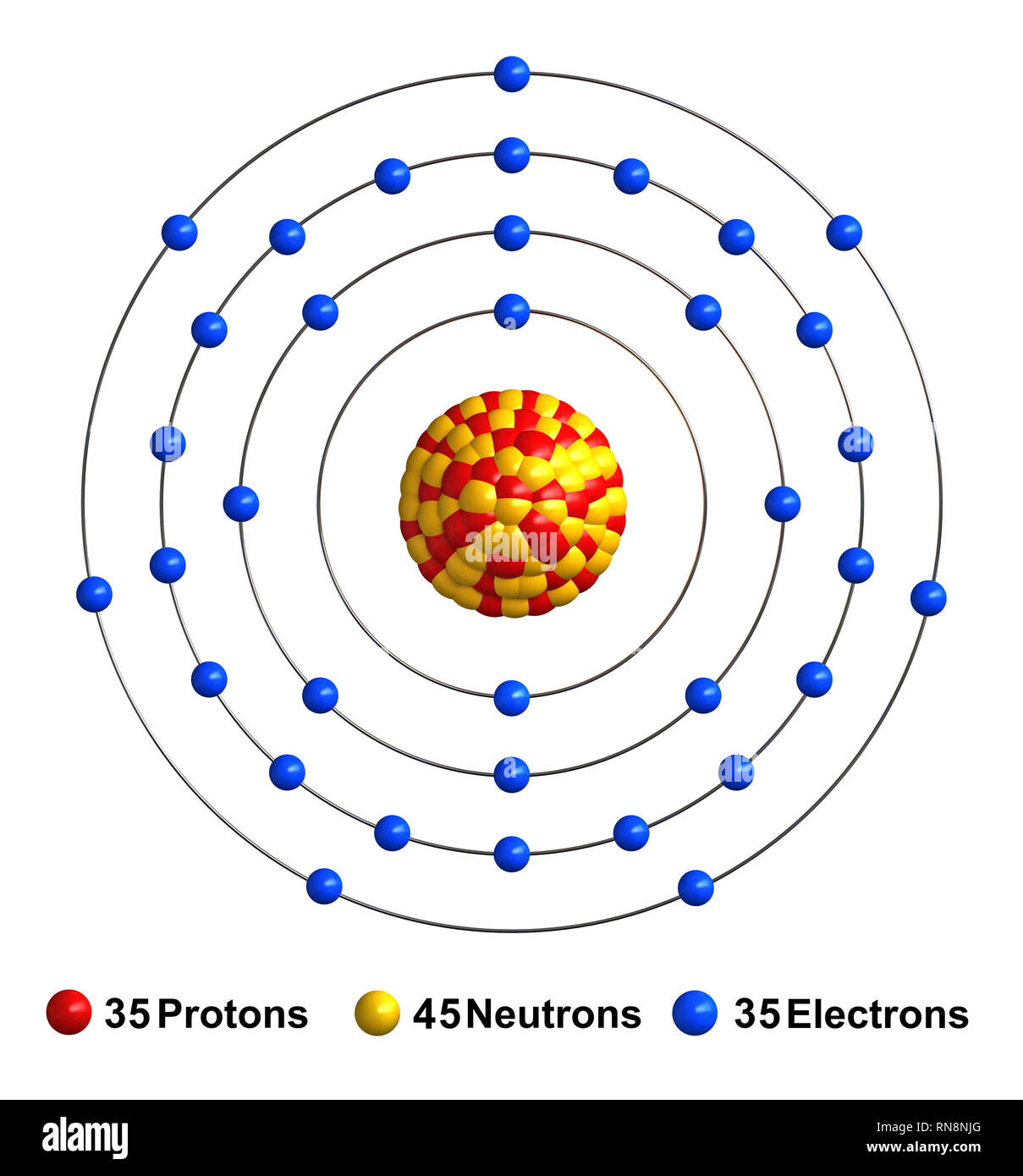
Basic Information Name: Bromine Symbol: Br Atomic Number: 35 Atomic Mass: 79.904 amu Melting Point: -7.2 °C (265.95 K, 19.04 °F) Boiling Point: 58.78 °C (331.93 K, 137.804 °F) Number of Protons/Electrons: 35 Number of Neutrons: 45 Classification: Halogen Crystal Structure: Orthorhombic Density @ 293 K: 3.119 g/cm 3 Color: Red Atomic Structure
Bromine Bohr Diagram
Immediately before 1913, the Rutherford model conceived of an atom as consisting of a tiny positively charged heavy core, called a nucleus, surrounded by light, planetary negative electrons revolving in circular orbits of arbitrary radii. Britannica Quiz Matter and More Quiz How does Niels Bohr's atomic model work?
Bromine (Br) AMERICAN ELEMENTS
In atomic physics, the Bohr model or Rutherford-Bohr model of the atom, presented by Niels Bohr and Ernest Rutherford in 1913, consists of a small, dense nucleus surrounded by orbiting electrons.
Numero De Electrones De Bromo lios
Figure \(\PageIndex{7}\) In Bohr's Model of the atom, electrons absorb energy to move to a higher level and release energy to move to lower levels. (CC BY-SA 3.0; Kurzon). The evidence used to support Bohr's model came from the atomic spectra. He suggested that an atomic spectrum is made by the electrons in an atom moving energy levels.

Bohr Model Bromine
From the Bohr model, it can be found that the number of orbits or shells in bromine is 4. Hence, as bromine has 4 orbits, it lies in period 4 of the Periodic table. Why is Bromine in p-block? Before knowing this reason, first of all I want to ask you a simple question. How can you determine the blocks-wise position of elements?
Bromine (Br) AMERICAN ELEMENTS
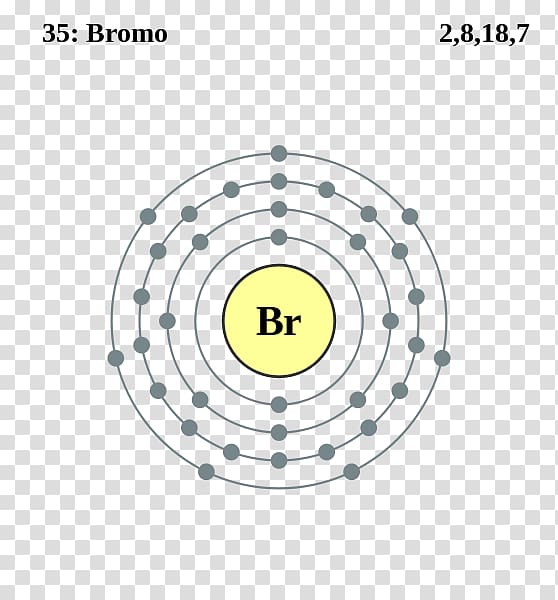
Bromine is a chemical element of the periodic table with chemical symbol Br and atomic number 35 with an atomic weight of 79.901 u and is classed as a nonmetal.. Bohr model: Electron shell for Bromine, created by Injosoft AB Br. Figure: Shell diagram of Bromine (Br) atom. Orbital Diagram. 1s: 2s: 2p: 3s: 3p: 3d: 4s: 4p:

Bohr Model Bromine Atom Electron Structure เวกเตอร์สต็อก (ปลอดค่า
Bohr Atomic Model of Bromine - Electrons per energy level. n s p d f; Ground State Electronic Configuration of Bromine - neutral Bromine atomAbbreviated electronic configuration of . BromineThe ground state abbreviated electronic configuration of Neutral . Bromine atom is [Ar] 3d10 4s2 4p5.
Electron Configuration Bromine Chemical Element Electron Shell Bohr
Expert Answer. Answer : 2-8-17-8. Solution :- According to Bohr model electron revolve around the nucleus in a circular path called orbit . The orbit can accomodate a maximum of 2n2 electron in their nth.. View the full answer.