Ethanol, Ethyl Alcohol Molecule, Chemical Structure. Skeletal Formula
Lewis Structure For Ethanol
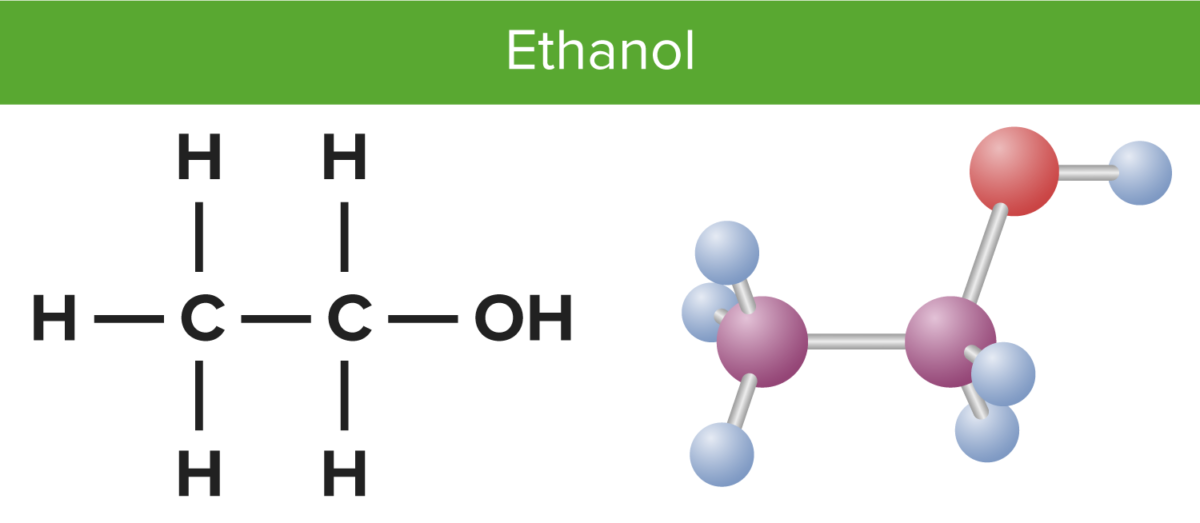
Ethanol Lewis structure November 2, 2023 by Deep The information on this page is fact-checked. Ethanol Lewis structure CH 3 CH 2 OH or C 2 H 5 OH or C 2 H 6 O (ethanol) has two carbon atoms, six hydrogen atoms, and one oxygen atom. In the ethanol Lewis structure, there are five C — H bonds, one C — C bond, one C — O bond, and one O — H bond.

Lewis Structure For Ethanol
Lewis structure is a representation of all the bonds and lone pairs of different atoms that a compound has. This is a 2-D representation and it helps us to understand more about the properties of the compound. Let's move step-by-step and see how the Lewis Structure of C2H5OH can be made. Step 1: Finding the valence electrons for each atom.
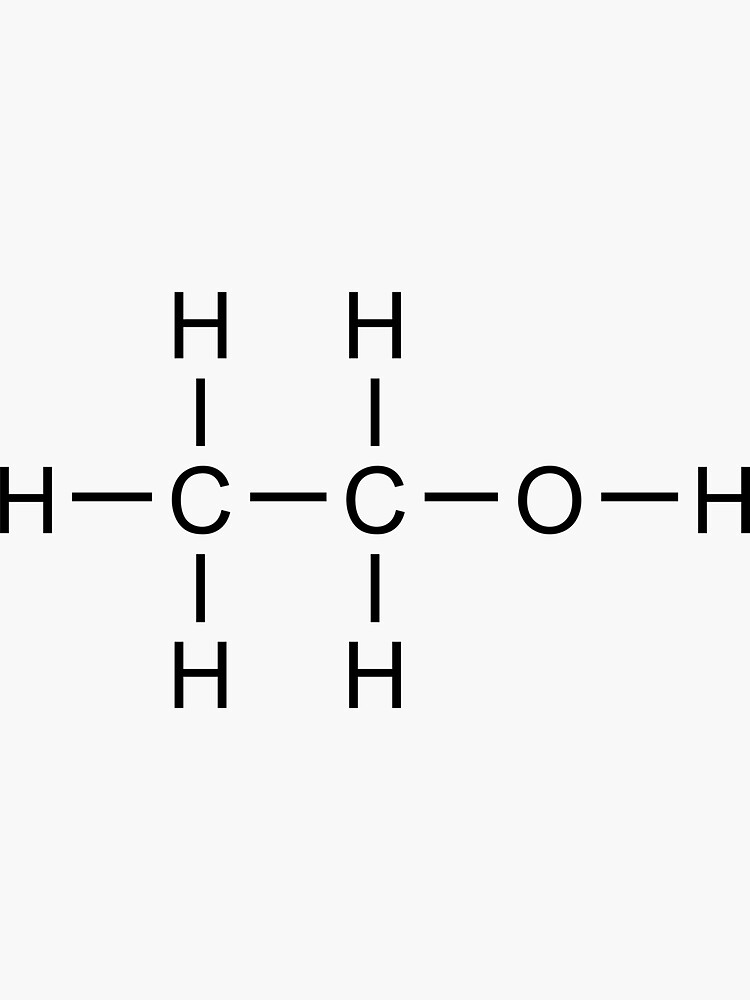
"Ethanol (Alcohol) Structural Formula" Sticker for Sale by
Steps of drawing C2H5OH lewis structure Step 1: Find the total valence electrons in C2H5OH molecule. In order to find the total valence electrons in a C2H5OH molecule, first of all you should know the valence electrons present in carbon atom, hydrogen atom as well as oxygen atom. (Valence electrons are the electrons that are present in the outermost orbit of any atom.)

Schema De Lewis De Ethanol
Molecular weight: 46.0684 IUPAC Standard InChI: InChI=1S/C2H6O/c1-2-3/h3H,2H2,1H3 IUPAC Standard InChIKey: LFQSCWFLJHTTHZ-UHFFFAOYSA-N CAS Registry Number: 64-17-5 Chemical structure: This structure is also available as a 2d Mol file or as a computed 3d SD file The 3d structure may be viewed using Java or Javascript . Isotopologues: [2H6]ethanol

Ethanol Formula Composition, Uses, Structure, Density
The Lewis structure is a simple yet powerful tool for understanding the bonding and arrangement of atoms in a molecule. In ethanol, we have a combination of carbon (C), hydrogen (H), and oxygen (O) atoms. To construct the Lewis structure, we start by counting the valence electrons. Carbon has 4 valence electrons, oxygen has 6, and hydrogen has.
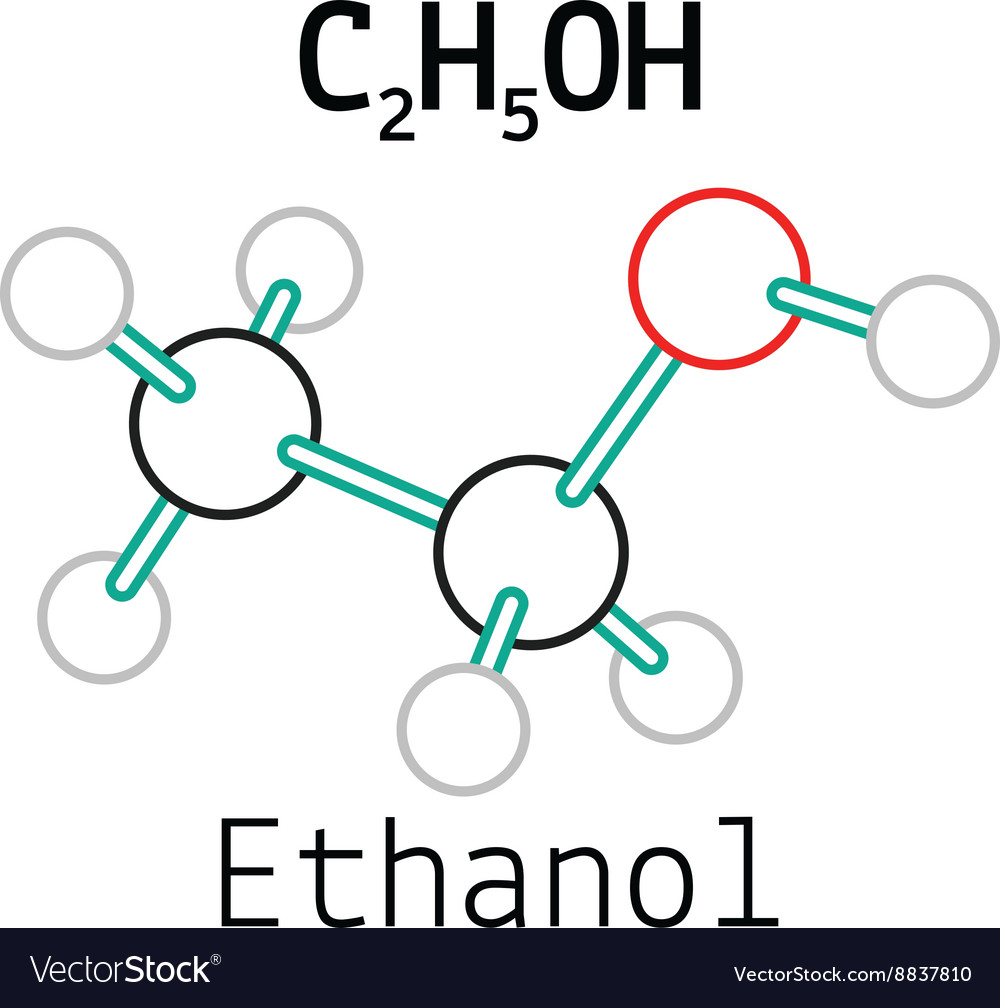
C2h5oh ethanol molecule Royalty Free Vector Image
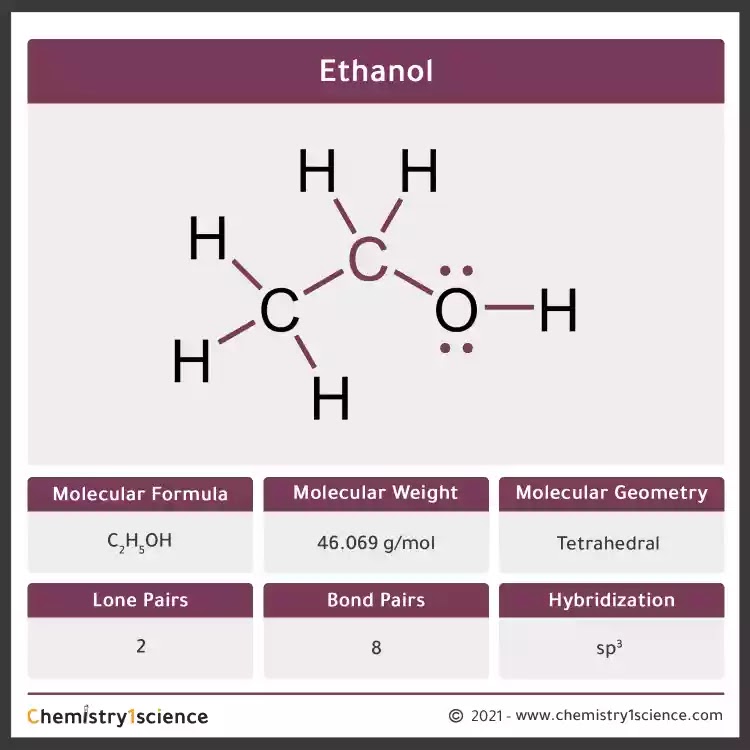
The Lewis structure of ethanol helps us understand its physical and chemical properties, including its molecular geometry, bond angles, polarity, and reactivity. The molecular geometry of ethanol is tetrahedral, with the oxygen atom at the center and the carbon and hydrogen atoms arranged around it. This geometry is a result of the.
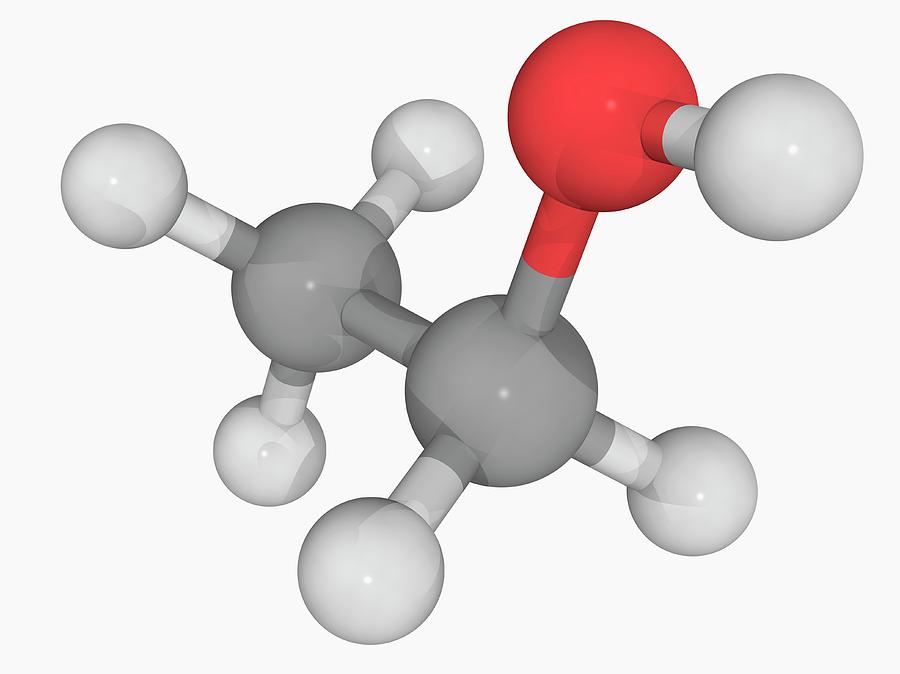
Ethanol Molecule Photograph by Laguna Design/science Photo Library Pixels
Get the free "Lewis Structure Finder" widget for your website, blog, Wordpress, Blogger, or iGoogle. Find more Chemistry widgets in Wolfram|Alpha.
A Look At The Ethanol Lies
The Lewis structure for ethyne, a linear molecule, is: The IUPAC nomenclature for alkynes is similar to that for alkenes except that the suffix -yne is used to indicate a triple bond in the chain. For example, CH 3 CH 2 C ≡ CH CH 3 CH 2 C ≡ CH is called 1-butyne.

Ethanol Formula Formula, Composition, Uses, Structure Embibe
Its molecular formula is C2H5OH, and it consists of two carbon atoms, six hydrogen atoms, and one oxygen atom. The Lewis dot structure of ethanol shows the arrangement of these atoms and their valence electrons.
/GettyImages-136810090-56a133b25f9b58b7d0bcfd93.jpg)
Ethanol Molecular Formula and Empirical Formula
Step 1: Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. When drawing the structure of an ion, be sure to add/subtract electrons to account for the charge. Step 2: Connect the atoms to each other with single bonds to form a "skeleton structure.".
Ethanol Metabolism Concise Medical Knowledge
Ethanol and its History. In the mid 1800's the use of grain alcohol (ethanol) increased as a major source of lamp fuel. Gas Lamp. Unfortunately, there were taxes placed on alcohol to help fund the.

Is Ethanol (C2H5OH) Polar or Nonpolar? Techiescientist
An alcohol is an organic compound with a hydroxyl (OH) functional group on an aliphatic carbon atom. Because OH is the functional group of all alcohols, we often represent alcohols by the general formula ROH, where R is an alkyl group.Alcohols are common in nature. Most people are familiar with ethyl alcohol (ethanol), the active ingredient in alcoholic beverages, but this compound is only one.
Ethanol Molecule Alcohol Universe Chemistry PNG, Clipart, Alcohol
Lewis structure of C2H5OH (or Ethanol) contains five C-H bonds, one O-H bond and one C-O bond. The two Carbon atoms (C) are at the center and it is surrounded by Hydrogen atoms (H) and one OH group. The oxygen atom has 2 lone pairs. Let's draw and understand this lewis dot structure step by step. (Note: Take a pen and paper with you and try.
Ethanol Molecular Geometry Hybridization Molecular Weight
How to Draw a Lewis Structure for Ethanol? C2H6OLewis Structure: https://www.youtube.com/watch?v=4rRVPeeZRmc&list=PLDwv-O7TJyNjAB0ak6We0sQ8t_a7D2cJ7Subscribe.
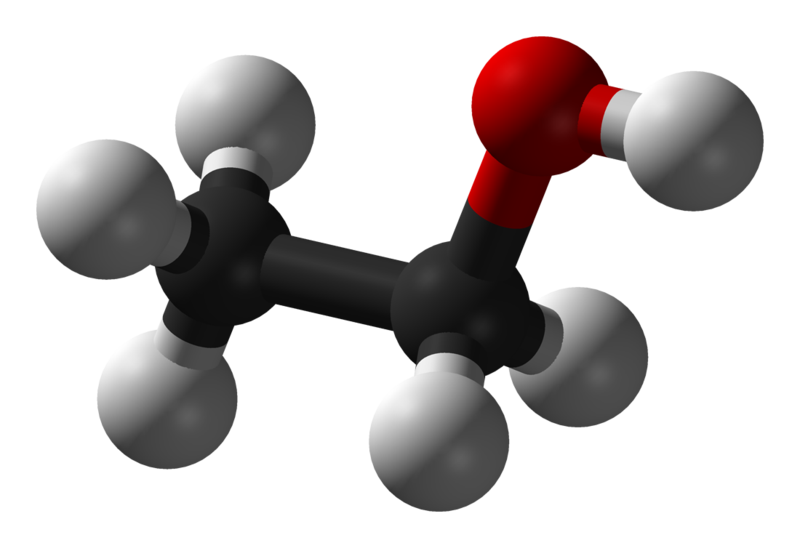
Ethanol molecule c2h6o modeled 3D model TurboSquid 1543644
C2H5OH or Ethanol is an organic chemical compound, which can also be represented as CH3-CH2-OH. Ethanol is a colourless liquid with a distinct odour and has a pungent taste. It has flammable properties; and gives a blue colour flame when burnt.

Ethanol Formula Formula, Composition, Uses, Structure Embibe
Lewis Dot Structure of CH3CH2OH (Ethanol) kentchemistry.com 25.1K subscribers Subscribe Subscribed 80K views 12 years ago Every Video I quickly take you through how to draw the Lewis Structure.